The curvature of membranes can also enrich proteins of certain permissible sizes and mechanical properties. The phosphorylated tyrosines attract proteins possessing SH2 (src homology) or PTB (phosphotyrosine binding) domains, which comprise a signaling complex. For example, Receptor Tyrosine Kinases (RTKs) phosphorylate certain tyrosine residues in the cytosolic domains of transmembrane proteins upon ligand binding to the extracellular domains. Īdditionally, cytosolic domains of transmembrane proteins can attract other proteins present in the cytosol that participate in signaling cascades. It is also suspected to induce local actin depolymerization in Dictyostelium cells and influence cell migration although other studies in neutrophils have indicated its role is in actin polymerization. The enrichment of PIP3 has been shown to accumulate at tips of neurons thus inducing cell polarization and axon formation. Another closely related phosphoinositide, Phosphatidylinositol 3,4,5 triphosphate (PIP3) may also be enriched in certain regions of the plasma membrane. FERM domain containing proteins, that link actin with the membrane, can also interact with PIP2. PIP2 is enriched in certain regions on the membrane, and due to its ability to interact with the PH (pleckstrin homology) domains of proteins it can locally recruit proteins possessing such domains. Phosphatidylinositol 4,5 bisphosphate (PIP2) is the most abundant phosphoinositide in mammalian cells. Ĭytosolic proteins that are associated with the plasma membranes are often localized based on their interaction with membrane lipids such as the phosphoinositides. For example, apical membrane proteins are very often anchored to GPI (glycophosphatidylinositol) while basolateral proteins have diLeu (N,N-Dimethyl Leucine) or tyrosine amino acid based signature sequences. This is achieved through the recognition of distinct signal sequences that target proteins to each of these regions. For example, in epithelial cells, which are polarized, protein composition at the apical membrane is very different from that at the basolateral membrane. Interestingly, the organization of a cell, and its various regions, do play a role in directing the recruitment of proteins to a given site. This usually consists of about 30 amino acids that are not present in a linear sequence, but are in close spatial proximity in the 3-dimensional space. In other cases proteins may carry a signal patch. Proteins that are destined for the endoplasmic reticulum also carry a signal peptide. The nuclear localization signal is one such example. For example, many membrane bound proteins carry signal peptides that are recognized by signal receptors that guide them to the target site. Protein recruitment is essentially a form of protein recognition, made possible by the presence of specific amino-acid sequences within the protein structure. Usually, signal sequences are cleaved off co-translationally by SPases (signal peptidases), and the resulting cleaved signal sequences are termed signal peptides (4-5). 
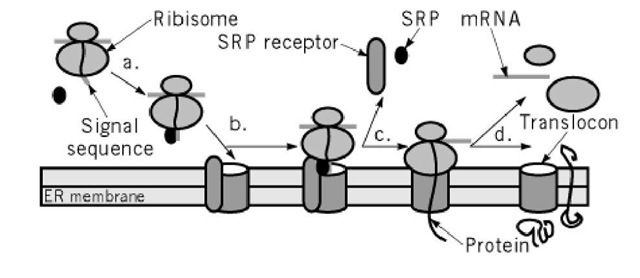
The growing peptide is gradually transferred via the protein-conducting channel (the translocon) into the ER lumen (4). Following the complex positioning (3), SRP / SRP receptor complex dissociates from the peptide due to GTP hydrolysis. 
GTP binding to SRP and SRP receptor is a prerequisite for SRP / SRP receptor complex formation. In eukaryotes peptide elongation pauses upon SRP / ribosome nascent chain complex formation the complex is then targeted to the ER membrane by the interaction with the SRP receptor (3). SRP (signal recognition particle) interacts with the signal sequence as soon as it emerges from the ribosomal polypeptide exit tunnel (1-2).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
